There have been some interesting discussions on the F.E.A.S.T. Facebook group over the past month regarding the role of genetics, personality traits, environmental factors and their role (or lack thereof) in the development of eating disorders and their prognosis. A parent group may seem like an unlikely forum for several hundred-odd comment threads on etiology; however, what we (caregivers, patients or clinicians) believe to underlie these disorders naturally informs our attitudes, decisions and choices with regards to treatment and our relationship to the disorders themselves:
Is this something they will have to manage their entire life?
Does anyone ever fully recover?
I had bulimia as a young adult and now my son has an eating disorder, too – did I pass on “bad genes”, bad habits, or is it a coincidence?
Is her rigidity and anxiety merely a side affect of starvation, or should we treat those as an underlying factor in her food refusal?
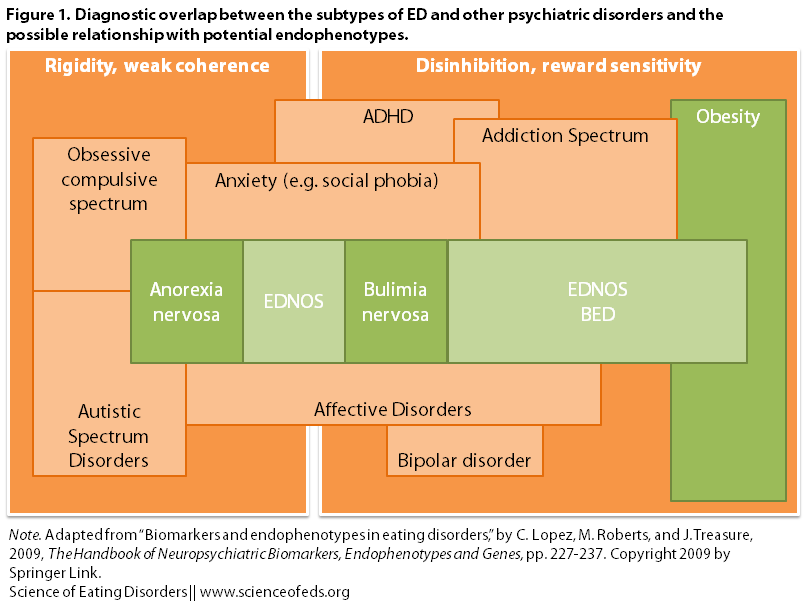
Is there any validity to the stereotyping of anorexics as uptight, overachieving perfectionists, and bulimics as impulsive, uninhibited hedonists?
There is at present no Grand Unified Theory of why someone develop an eating disorder and, despite what pop science reporting would have us believe, there is no “gene for” AN, BN, BED or ED-NOS and no genetic or physiological test for diagnosis. Even the terminology we use to describe eating disorders is subject to contention: are they a “brain disorder“, a “biologically-based mental illness” or a psychiatric disorder? (And what is the difference?)
An instrumental part of science literacy, in my opinion, is the ability to get down and comfortable with uncertainty: Well, it looks like this might be be the case, but we don’t yet have enough evidence to say so definitively. And the ability to appreciate that two seemingly disparate or contradictory ideas can both be true. Research Methods 101: A theory can never be wholly proved, only disproved. (I won’t get into the the “file drawer effect” – why there’s a bias against publishing null (p>.05) findings and why replicating results from prior studies does not occur nearly as often as it ought to – but you can read more about the issue here.) Human beings are infinitely complex; thus, so, too, are our problems.
Recent work in the field aims to clarify biological contributions to the maintenance and development of eating disorders, as well as the role of biology in conditions that are commonly co-occur with EDs or in their family members: e.g., anxiety disorders (obsessive-compulsive disorder, generalized anxiety, social anxiety); mood disorders (major depression, bipolar disorder); autistic-spectrum disorders (Asperger’s); attention-deficit disorders (ADD, ADHD) and substance use and addiction.
To explore this phenomenon, researchers often look at potential biomarkers and endophenotypes. This post will focus on a chapter by Carolina Lopez, Marion Roberts, and Janet Treasure from The Handbook of Neuropsychiatric Biomarkers, Endophenotypes and Genes (2009).
A biological marker, or biomarker, is measurable physical evidence, the presence, amount or absence of which indicates either a disease state, normal physiological functioning, or response to treatment. Often this evidence is a substance (such as a specific protein) in the blood or tissues, but biomarkers can also be neuropsychiatric or neuropsychological. These biomarkers can be measured by imaging techniques that allow us to glimpse physical changes in the brain, or by performance on standardized assessments that aim to measure disease features like memory deficits. (Here is a model of the different functions that biomarkers can fulfill.)
Biomarkers do not necessarily have a genetic basis.
Endophenotypes are heritable observable characteristics or behavioral traits that may “underlie and contribute to certain disease vulnerabilities but [are] not…part of the disorder itself.” Endophenotypes may involve multiple genes, but attempting to establish a genetic basis for a single one of these underlying traits is much less complex than trying to delineate the genetics of an entire disorder. In their 2003 paper, Gould and Gottesman propose several criteria for endophenotypes:
1. The endophenotype is associated with illness in the population.
2. The endophenotype is heritable.
3. The endophenotype is primarily state-independent (manifests in an individual whether or not illness is active).
4. Within families, endophenotype and illness co-segregate [meaning they are inherited together].
Subsequently, an additional criterion that may be useful for identifying endophenotypes of diseases that display complex inheritance patterns was suggested:
5. The endophenotype found in affected family members is found in nonaffected family members at a higher rate than in the general population.
In the chapter, Lopez et al. discuss the difficulty of ascertaining potential biomarkers in the ED population, due to the profound effects that the behaviors have on the individual’s body and mind. Some attempt to circumvent this issue by looking at those who are recovered from their ED, but that method is also problematic, for two reasons. First, there is no consensus on how to define “recovered.” Second, researchers cannot be certain that they are observing true biomarkers – they may be instead what the authors call “scar effects” from the disorder: perhaps a history of starvation, weight changes, or binge/purge behavior provokes changes in one’s physiology or psychological makeup that persist even after normal nutrition and body weight are restored.
Because few studies so far have examined biomarkers that represent a heritable genetic underpinning (and fulfill all criteria for a full endophenotype), the authors use the term “biomarkers” in their review. They propose two categories of biomarkers: those “those derived from conceptual and theoretical models of the illness, and those that result from strong empirical grounding.”
It is important to note that these categories are not necessarily mutually exclusive: this line of investigation is still in its infancy and a biomarker from the former group may migrate into the latter as we figure out how best to measure and obtain empirical evidence for the more theoretical constructs.
However, the chapter is focused primarily on the evidence-based biomarkers, which I will summarize in a two-part post: the first dealing with biomarkers involving neurotransmission, and the second focusing on neuropsychological biomarkers. You can read on for Part 1, or skip ahead to Part 2.
NEUROTRANSMITTER BIOMARKERS
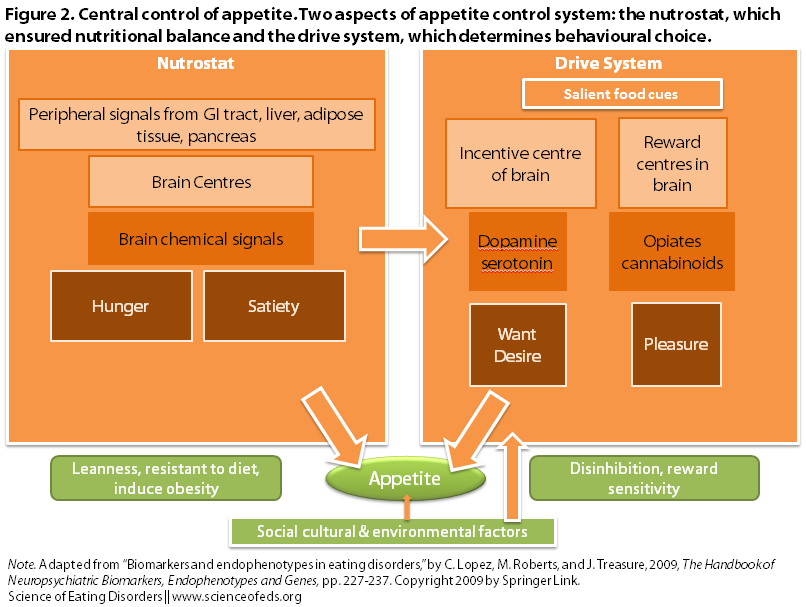
Changes in the neurotransmission systems for serotonin (5-HT) and dopamine (DA) can affect the processes in the brain and gut that regulate appetite and eating (as well as reward and emotion processes):
Lopez et al. state: “A number of anomalies in the serotonin (5-HT) system have been described as enduring features in ED.” These are represented in the table below, along with the changes seen in the dopamine system:
Interestingly, they note that the increase in dopamine receptors (resulting in less dopamine hanging out in the space between neurons – for comparison, SSRI and SNRI antidepressants like Prozac or Effexor work by inhibiting the receptors for neurotransmitters, meaning that more of that neurotransmitter hangs out in the synaptic space) in anorexia nervosa may explain the difficulty that people with AN (past or current) have in distinguishing positive and negative feedback, which in turn may make them less sensitive to things that most people find rewarding or pleasurable…such as eating.
(For a more comprehensive discussion of the neurotransmitter changes seen in EDs, check out Tetyana’s posts on serotonin function in bulimia nervosa and in restricting-type anorexia.)
Continue to Part 2: Neuropsychological Biomarkers
References
Carolina Lopez, Marion Roberts, & Janet Treasure (2009). Biomarkers and Endophenotypes in Eating Disorders The Handbook of Neuropsychiatric Biomarkers, Endophenotypes and Genes, 227-237 DOI: 10.1007/978-1-4020-9464-4_16