I used to call them bingeing and purging marathons. If I binged and purged in the morning, chances were, I’d binge and purge throughout the day. The next time I’d eat, I was likely to end up–whether I wanted to or not–bingeing and purging. Not all individuals with bulimia nervosa binge and purge every day (or purge everything they eat, for that matter), but many do, and some binge and purge multiple times a day. In recovery, many people start by trying not to binge and purge before a certain time of the day–because once they binge and purge, it triggers a continuous cycle of bingeing and purging until they become to exhausted or otherwise end up going to bed.
I always wondered why that was, why was it so hard to keep a single episode of bingeing and purging from initiating a repeated cycle of bingeing and purging?
On days I didn’t binge and purge, my eating was fairly normal. I had little trouble eating a normal-sized meal, keeping it down, and recognizing when I was full. I wasn’t too hungry or too full. I knew when I was hungry and when I was full. But on days I’d binge and purge in the morning (because I have a whole day of eating ahead), everything food-related would become chaotic and confusing. Part of it, surely, was the “Well, I screwed up, so why not binge/purge some more” mentality, but part of it also felt physiological. I couldn’t really tell if I was hungry, or full. When I’d eat again, I couldn’t tell when to stop, even though I had no trouble knowing on days I didn’t binge and purge.
So, why did that happen?
In a paper published in 1994, Johnson and colleagues investigated the role of insulin and glucose as possible mediators of these repeated bingeing/purging episodes. The bingeing and purging marathons.
Insulin is a key hormone in carbohydrate metabolism, and it is also a strong appetite stimulant. Insulin is secreted during two phases: after we eat (postprandial phase) to help transport glucose into cells, and right before we eat or when we are in the presence of food that we are expecting to eat (cephalic phase).
Johnson et al. hypothesized:
Many women with bulimia nervosa are through to be below their set-point or regular weight, and thus may display the same exaggerated cephalic insulin response as the obese. Also, the act of vomiting effectively prevents caloric absorption in the presence of cephalic and postprandial insulin elevations and may produce an exaggerated hypoglycemia, the consequence of which may be the initiation of another binge/purge episode.
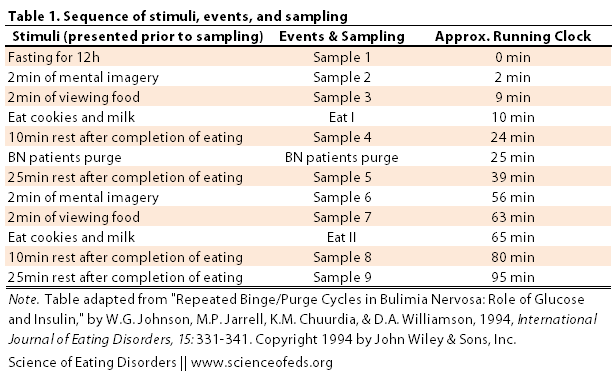
To test these hypotheses, Johnson et al recruited eight patients with bulimia nervosa and eight healthy controls (in their mid-late 20’s, BMIs in the “normal” range). After a 12 hour fast, the women arrived at the laboratory and proceeded to follow the protocol below:
For the “mental imagery” component, the women were instructed to relax, close their eyes, and concentrate on imaging a meal they’d like to eat, and then imagine eating that meal. A sample of blood was collected after two minutes of imagining a meal (Sample 2). The women were then presented with a test meal of cookies and milk, and after 2 minutes of viewing the food, another blood sample was drawn (Sample 3). The subjects were then told to eat as much as they wanted (the amount they ate was recorded) and 10 minutes after completion of eating, another sample was drawn (Sample 4). The bulimia nervosa patients were then allowed to purge (via self-induced vomiting), and 25 minutes after termination of eating (“Eat I”), another blood sample was drawn (Sample 5). The same protocol (minus the purging) was repeated again (Sample 6-9, Eat II).
During each blood sample collection, heart rate and rating of hunger, bloatedness, anxiety, and urge to purge were also collected to make sure that the laboratory settings mimicked a natural environment.
THE MAIN MAIN FINDINGS
Glucose
- Glucose levels following the 12h fast (Sample 1), prior to eating (Sample 2,3) and following “Eat I” (Sample 4) were comparable between the two groups
- Glucose levels following purging were significantly different between the two groups (Sample 6)
- Indeed, ~20 minutes after purging, the glucose levels in the BN group were the same as fasting glucose levels(~71mg/dL in BN group versus ~98mg/dL in control)
Insulin
- Insulin levels were comparable between groups throughout Sample 1-3, but were significantly higher in the BN group immediately prior to purging (this makes sense, to me, because the BN group ate roughly 2x more than the control group, so insulin likely spiked accordingly, though admittedly, I’m not sure about the lag between eating and insulin/glucose changes) [thanks to Natasha for pointing out my earlier mistake here]
- However, insulin levels were significantly lower in the BN group following purging (Sample 6 and 7)
- After eating the second meal (and not purging it), insulin levels were comparable (not different) between the two groups at Sample 9
Other Data
- BN subjects ate more during “Eat I” phase, and drank and eat more during the “Eat II” phase
- Heart rate was positively correlated with anxiety, bloatedness, and urge to purge (and negatively correlated with hunger)
- BN group had a higher heart ate and anxiety ratings through-out the experiment, compared to control group
- BN group experienced highest heart rate immediately prior to purging, and lowest (almost as low as control group) ~20 minutes after purging. Heart rate increased after “Eat II” in the BN group.
Johnson et al. concluded that the heart rate and self-reported anxiety, hunger, and bloatedness measures suggested that the laboratory experiences mimicked the “natural environment” (or typical bingeing and purging episode at home).
So what does the data on insulin and glucose suggest about the repeated binge/purge cycles?
First, the fact that there were no differences between control group and BN group after fasting, while thinking about food, and in the presence of food (Samples 1-3) suggests AGAINST that idea that increased insulin release prior to eating (cephalic phase) maintains the binge/purge cycles (first part of the hypothesis).
Second, the fact that insulin and glucose levels were comparable after “Eat II” (no purging) also suggests that postprandial insulin and glucose levels DO NOT maintain the binge/purge cycle.
The fact that insulin and glucose levels were normal in the absence of binge eating and purge is important because it strongly suggests that women with bulimia nervosa have, on the whole, normal metabolic/endocrine function.
Strikingly, however, glucose levels plummeted in bulimia nervosa patients following purging of the first meal to the same level seen following the 12 hour fast.
Low blood sugar (hypoglycemia) results in hunger and likely predisposes individuals to bingeing and purging again.
The authors conclude,
In summary, the absence of both an exaggerated cephalic phase insulin response and persistently high insulin levels following a purge fail to support a role for insulin in repeated binge/purge cycles. In contrast, the hypoglycemia resulting from purging is more likely to contribute to the continuation of repeated binge/purge episodes. […]
The abnormalities observed in some studies do not appear to represent a primary, aberrant biological process, but most likely the abnormal endocrine/metabolic findings are a consequence of alternating periods of semistarvation, binging, and vomiting. Moreover, bingeing and purging are behaviors with demonstrable physiological consequences, and our results suggest that hypoglycemia subsequent to purging plays a role in the continuation of binge/purge cycles.
Of course, there are a lot of limitations in this study: it is tiny, and despite the self-reported anxiety, hunger, and bloatedness levels, it is hard to confirm that the laboratory setting truly mimics a “natural” environment. Eating a few cookies and drinking two glasses of milk is much less than what most BN patients would consider a binge (though, I hesitate to define a binge based on the amount of food eaten). The values in the paper do not have standard deviations (which is very odd to me–I’m not sure what the explanation for this is), and it is hard for me to comment on the reliability of the methods used to measure insulin and glucose levels.
Still, though the findings don’t conclusive prove anything, they are suggestive and provide an attractive explanation to explain how a patient with bulimia nervosa might be able to eat fairly well some, or even most, of the time, and yet when an episode of bingeing and purging happens, quickly snowball into one, two, or more days of continuous repeated bingeing and purging episodes.
I’m not an endocrinologist, so if I got something wrong, please leave a comment. Also, I’d like to thank Saren for brining this paper to my attention.
EDIT: Just to be clear–there are a lot of other hormones that regulate hunger and satiety, so I don’t want to accidentally imply that this post-purge hypoglycemia is the only thing that occurs, it is just that this paper focused on investigating insulin and glucose specifically. Also, it is quite interesting that in the paper, the authors often used the term “bulimics”–not something you see now-a-days in the peer-reviewed literature (it is generally, “bulimia nervosa patients,” or “BN group,” or something like that). Interesting to see how things have changed in the last 20 years (for the better, in this regard).
References
Johnson, W., Jarrell, M., Chupurdia, K., & Williamson, D. (1994). Repeated binge/purge cycles in bulimia nervosa: Role of glucose and insulin International Journal of Eating Disorders, 15 (4), 331-341 DOI: 10.1002/eat.2260150404
Thanks for posting this. I found it very interesting. I am no scientist (indeed, I had to read it quite a few times over to make sense of it in my head!) and so apologies for what may appear a very dumb question… but what was the explanation given for the BN group’s insulin having been significantly higher than the control group’s immediately prior to “Eat I”?
Hey Natasha,
Not a dumb question at all and you have a very good eye *because* you noticed my mistake! What I meant to write was that the insulin levels were the same for sample 1,2, and 3. They were significantly higher immediately prior to purging.
So, before purging the insulin levels were higher in the BN group, and, while they don’t discuss this, it makes sense because the BN group ate roughly 2x more than the control group, so it makes sense for the insulin to spike up. After purging, the insulin levels in the BN group decrease and then continue falling for sample 5,6 and 7, whereas they increase a bit and stay relatively stable for the control group.
(I added ^ in the post.)
In sample 9, 25 minutes after eating (and not purging) the second meal, the insulin levels were the same between the two groups.
Thanks for pointing this out and I hope that clarifies everything?
Thanks so much for posting Tetyana! I hadn’t seen this 20yo study, but it is very helpful given that so many patients blame “willpower” for their inability to interrupt the binge/purge cycle.
I wanted to thank you for sharing this as well. I find the study every interesting! I have struggled with this Ed severely for over 20 yrs , and despite spending over half my life in treatment or hospitals and residential centers, I still struggle with the bulimia very much, and the way you described it is exactly what I experience. I feel like the longer I put off eating at all each day, the safer I will be… But all of the foods I have been choosing to eat are binge foods …so it is always a constant and expensive daily struggle. I have actually been trying, on my own, to delve more into the physiology and studies done which look at different hormones, glucose, etc … As if once I figure it out perfectly , I can cure myself. Ugh…if only! But I really did appreciat this article and read it twice…with great interest. Thanks again, and best of luck to you and continued recovery!
Debbie, I’m glad you found it useful! I too always hoped that knowing the neurobiology and physiology would help me with recovery (I don’t think it did, but it did make me more aware of the physiological underpinnings of my behaviours and enabled me to feel less blame and guilt). I know a lot of people that try to procrastinate eating because of fear that once they start they’ll binge. Unfortunately, the only way out of it is to actually start eating early and through-out the day. Even just a bit, and even “safe” foods. It is vital, otherwise there’s really no way out of the terrible cycle. What was hard for me was that non-triggering foods were a pain in the butt to digest and led to so much abdominal discomfort sometimes that I’d purge because I couldn’t deal with the pain. :/ (I don’t struggle with that at all anymore, but it was terrible.)
Best of luck to you with your recovery.
Tetyana
Hello. I thank you immensely for thss article as was quite informative and rather intriguing to the point of satisfaction. I have suffered for the past three years, with EDNOS but it seems to becoming worse. Honestly, before I used to fast but its becoming closer to bulimia nervousa. I’m 16 and I feel as if the tub yearns for my food…but the strange thing this article brought to my notice was that when I binge and purge it happens in continuous cycles over and over again. Also I have gotten hypoglycemia symptoms much more often now when before I used to fast for weeks at a time. I don’t understand the sudden increase.
I think this is very helpful and interesting. I know my glucose levels have dropped so severely after purging that I would have hallucinations and seizure-like tremors. Then I would binge on sweets again just to get my blood sugar back up.
Thank you so much for this. I have suffered from bulimia since 2007. My experience has been, as others have said, no problem with food on some days, know when hungry, not hungry, but when I start a binge/purge I can not stop. Often buying food binges 10 times a day. It feel very physiologically driven once it starts and I get very frustrated as I wish the physicians who treat and research bulimia could experience one day of the physiological impacts it creates and how that contributes to the ong going nature of the illness. I believe science could definitely help reduce the impact if they researched more on this area.
“I wish the physicians who treat and research bulimia could experience one day of the physiological impacts it creates and how that contributes to the ongoing nature of the illness.” I agree! Unfortunately, I haven’t seen much research into the physiological drives :/.