There is this prevalent myth on Tumblr eating disorder blogs that increased dopamine (DA) receptor activity or increased DA signalling causes anorexia nervosa. It has left me quite perplexed, as I have never come across a single paper that has shown increased DA activity causes anorexia nervosa. My research for this post also left me empty-handed. I have no idea where this myth comes from, but I thought I’d blog about what research on DA activity in anorexia has shown. This topic will take me a few (not necessarily successive) posts to cover. This first post is a very brief introduction to DA signalling.
First, what is dopamine?
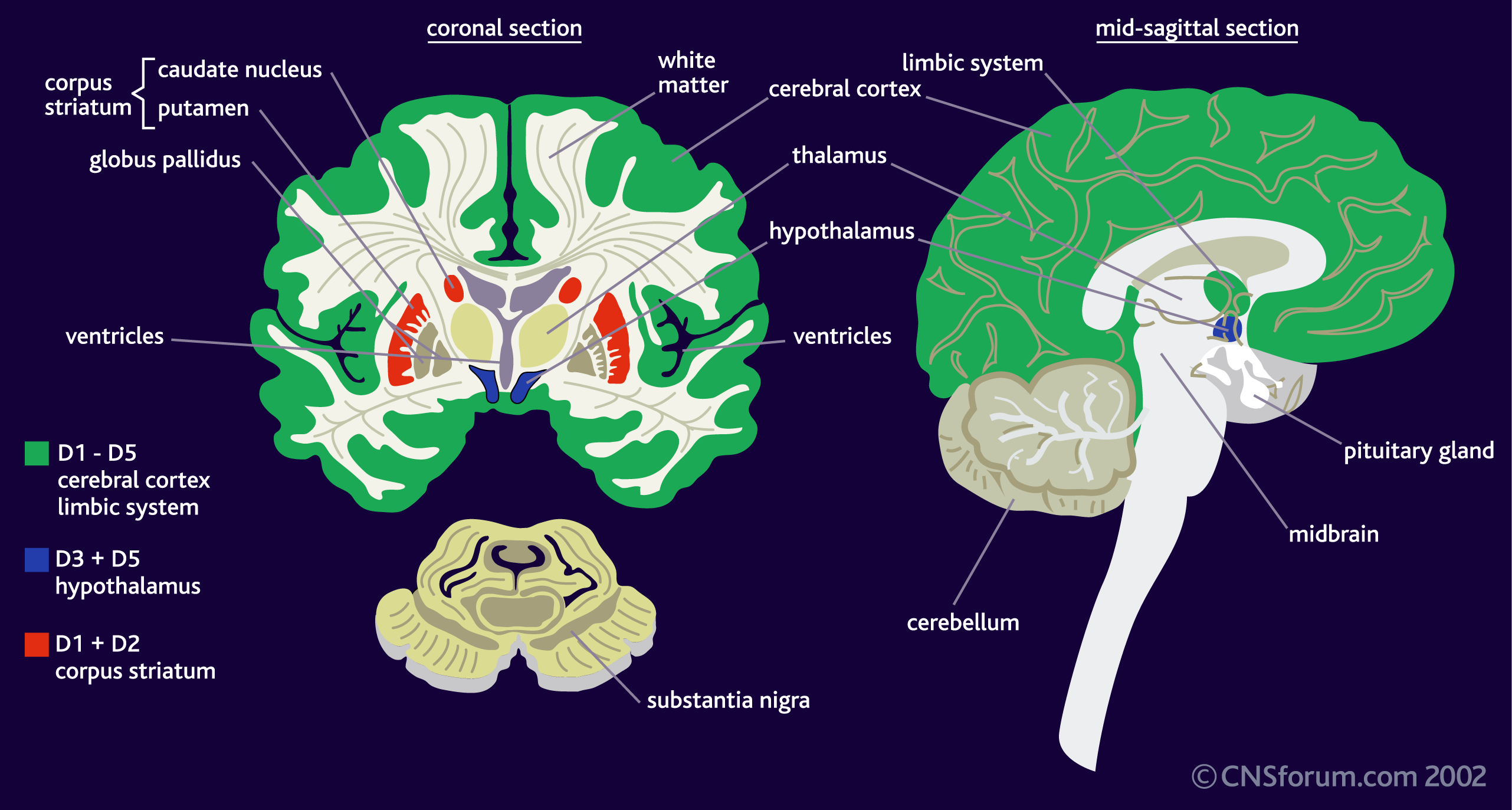
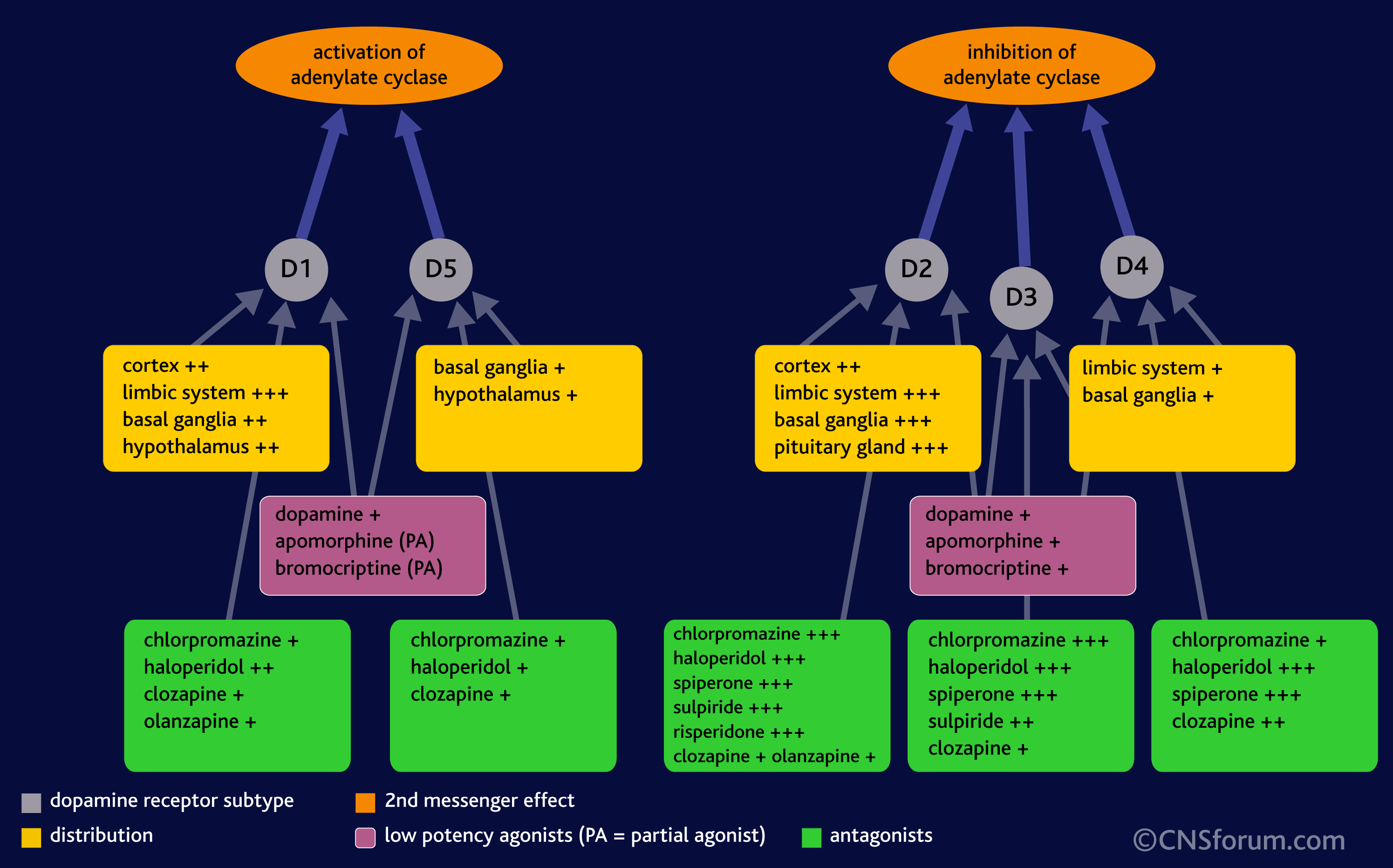
DA is a neurotransmitter–a molecule that one neuron releases to another in order to send a signal. Dopamine is released from one cell (the presynaptic neuron) and binds to its receptors on the other (postsynaptic) neuron. There are five different DA receptors in the human brain, called D1, D2, D3, D4 and D5. (To add to the complexity, there is often more than one version of the receptor, called an “isoform.”) The receptors have different (but overlapping) patterns of expression in the brain (areas where the receptors can be found):
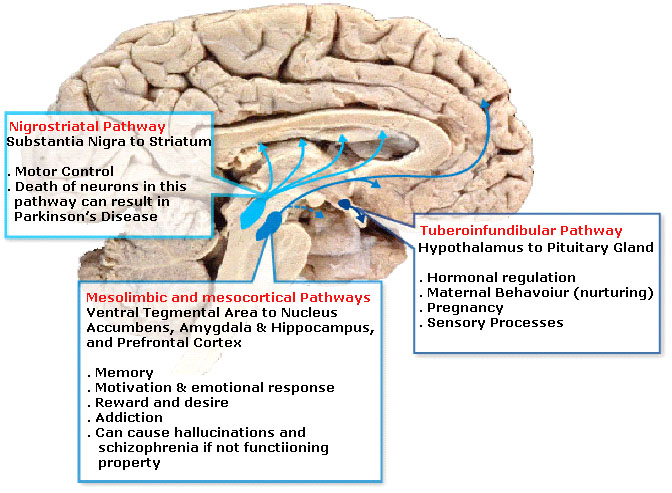
There are also several dopaminergic pathways (a neuronal pathway connects neurons from one brain region to neurons in another brain region), which are also involved in different aspects of behaviour. The main ones are described in the image below:
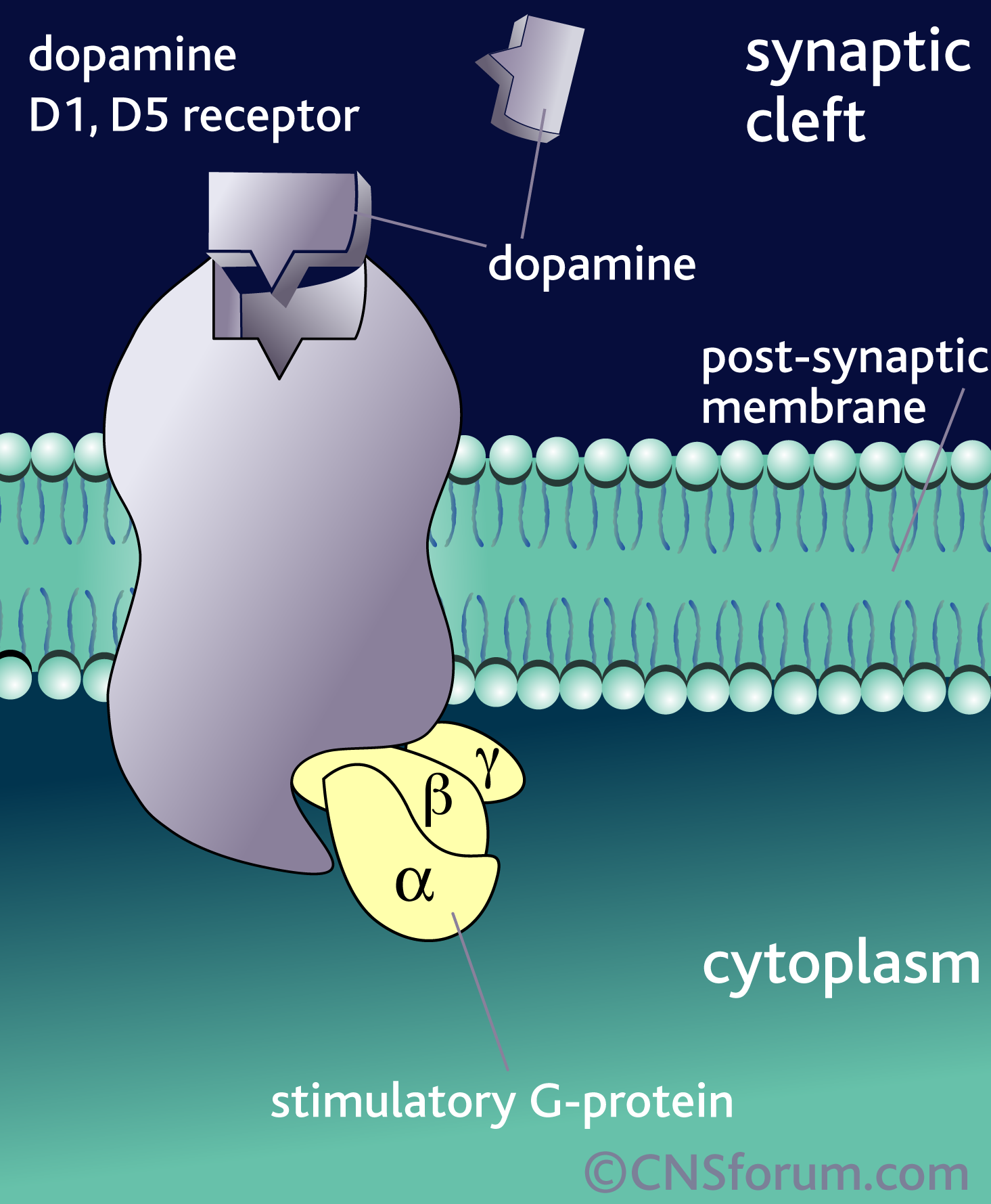
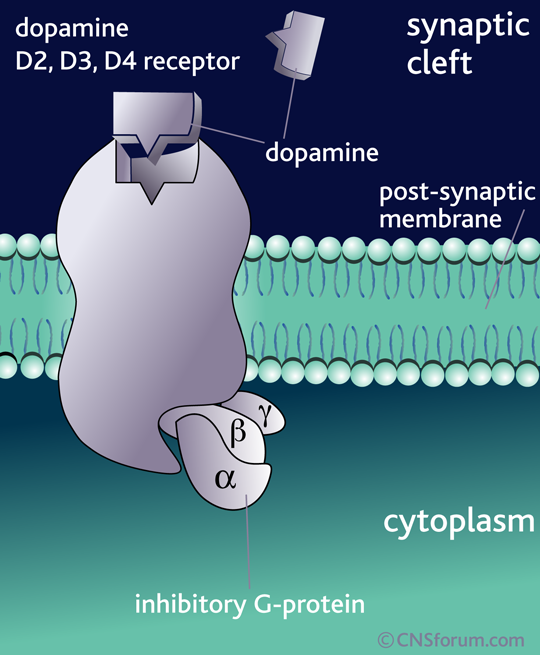
What’s important is not so much that DA is released but the specific receptor it binds to and the brain region that receptor is found in. The effect of DA release is thus dependant on the receptor, because it is the receptor that determines how the DA signal is perceived. For example, when DA binds to D1 or D5 receptors, it leads to the activation of a signalling molecule called adenylyl cyclase (also adenylate cyclase, or AC), but DA binding to receptors D2 through D4 leads to the inhibition of adenylyl cyclase:
Moreover, drugs are typically more potent inhibitors (antagonists) or activators (agonists) of some receptors more than others. That’s because although all of the receptors bind DA , they are still shaped slightly differently, and thus some drugs are more likely to bind to some receptor over others. For example, in the image below, you can see that chlorpromazine (thorazine or largactil) strongly inhibits D2 and D3 DA receptors when compared to D1, D4, and D5 receptors.
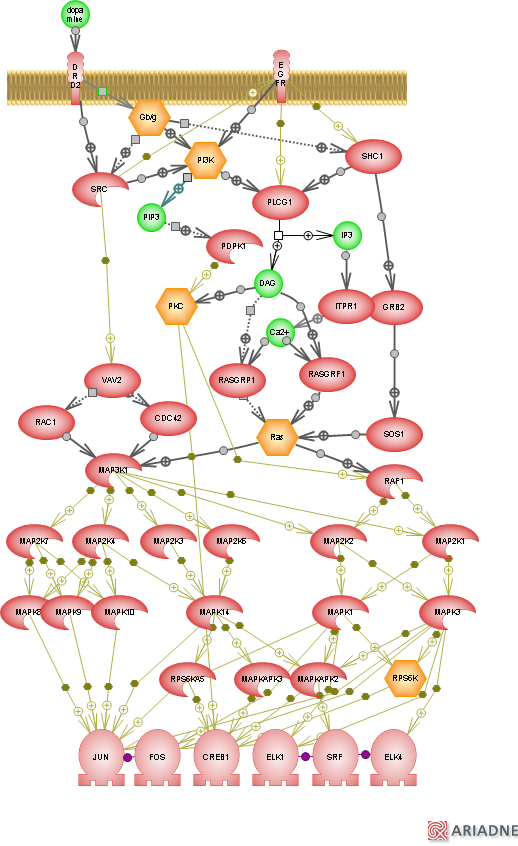
After DA binds to a receptor on the postsynaptic neuron, it activates a signalling cascade that can look pretty complicated. Signalling cascades are so complicated that studying the role of one protein in a specific brain region under a specific condition (like spatial learning) can take up the entire length of a PhD (or more). Here is just one example of a signalling cascade that occurs when DA binds to a D2 receptor (and there are many more):
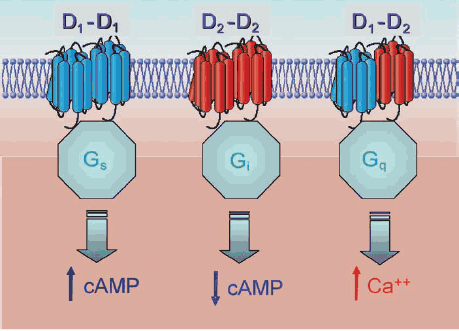
In addition, it seems that different types of DA receptors can come together to form a complex and further increase the complexity of DA signalling:
In one case, DA binding leads to an increase in a molecule called cAMP (cyclic AMP) but in the other, it leads to a decrease. In the third case, it leads to an increase in calcium levels (which has lots of implications for neuronal activity).
Hopefully this little introduction on dopamine signalling illustrates the complexity of this system. I hope that it is clear that what happens in the brain when DA is released really depends on a lot of factors, including (but not limited to), the receptors it binds to and the region of the brain those receptors are found in.
Dopamine is not a “reward” molecule, it is much more than that. In the central nervous system, DA is involved in “voluntary movement, feeding, affect, reward, sleep, attention, working memory, and learning. In the periphery, [DA] plays important physiological roles in the regulation of olfaction (smell), retinal processes, hormonal regulation, cardiovascular functions, sympathetic regulation, immune system, and renal functions, among others” (reviewed in Beaulieu & Gainetdinov, 2011).
Unsurprisingly, DA dysfunction is also associated with many diseases and disorders (from being at the very of the disease pathology to being potentially implicated in the disease), including Parkinson’s disease, schizophrenia, ADHD, Tourette’s syndrome, Huntington’s disease, drug abuse and addiction, bipolar disorder, major depression, dyskinesias, hypertension and kidney dysfunction (reviewed in Beaulieu & Gainetdinov, 2011).
Statements like dopamine receptor overactivity leads to anorexia nervosa are not just incorrect (because, as you’ll see in the next few posts, this has not been demonstrated) but also overly simplistic (even if the precise dopamine-anorexia nervosa link was clearly shown.)
In the next post, I’ll start delving into some of the research on the role of dopamine in anorexia nervosa. As a teaser, I’ll leave you with this quote from a recent review article I read on the topic (Kontis & Theochari, 2012):
There are at least two main [competing] theoretical models explaining the role of dopamine in AN. The first suggests that food restriction in AN is rewarding. [The second model] proposes that the reduction in food consumption observed in AN is because of a lack of pleasure associated with food intake… These two models fail to recognize the different aspects of reward and motivation and may be too simplistic. However, they could be a good starting point for a more detailed study of the role of dopamine in AN.
READ PART II (PRECLINICAL TRIALS with ANIMAL MODELS) HERE.
READ PART III (CLINICAL TRIALS with AN PATIENTS) HERE.
READ PART IV (TREATMENT with ANTIPSYCHOTICS) HERE.
PS. If you found any mistakes in this post, please kindly let me know. Likewise, if you found anything confusing or want me to elaborate on anything, again, please leave a comment below.
References
Beaulieu, J., & Gainetdinov, R. (2011). The Physiology, Signaling, and Pharmacology of Dopamine Receptors. Pharmacological Reviews, 63 (1), 182-217 DOI: 10.1124/pr.110.002642
Kontis, D., & Theochari, E. (2012). Dopamine in anorexia nervosa. Behavioural Pharmacology, 23 (5 and 6), 496-515 DOI: 10.1097/FBP.0b013e328357e115
….and then there are GABA circuits……
Well yes, of course, there are all sorts of neurotransmitter circuits. I blogged a tiny bit about serotonin in AN and BN, but haven’t gone into depth about any other NTs. I mainly want to do this series because time and time again I run into this thing on tumblr with someone claiming that increase dopamine receptor activity *CAUSES* anorexia, and it is just strikes me as a very random thing to say, given the lack of evidence.
Also, there’s not that much research on GABA and EDs. Do you have any specific papers in mind?
Here’s one:
Genetic association of recovery from eating disorders: the role of GABA receptor SNPs
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3176559/
Hi Millie, thanks.
I saw that paper when I did my search, but I don’t want to a SNP association study paper for the simple reason that there have been very many that contradict each other. Until there is more evidence from other groups that show the same association with the SNP, it is a preliminary finding and reporting a prelim finding might make readers think this is a “real” finding.
This is why I prefer to do review papers for association studies. This is taken from a review I wrote in my third-year of undergrad on similar types of studies but for a different SNP, to illustrate my point:
“Although there is evidence from brain imaging studies and psychiatric comorbidities that altered 5-HT neurotransmission is important in AN psychopathology, evidence pointing to specific gene polymorphisms has been contradictory. Certain alleles may result in altered 5-HT activity and may also underlie the high heritability of AN. Most attention has been given to the 1438A/G promoter polymorphism in the 5-HT2A receptor and so far out of 14 studies, six found an increased incidence of the -1438A polymorphism in AN patients [71-76], but eight others did not confirm the result [77-84]. Gene polymorphism studies of 5-HTT have yielded similar contradictory results, with some positive studies suggesting an association [85-88], and others showing no difference between AN and controls [76, 77].”
I don’t want to report on that one that found a positive correlation, when there will be, likely, several others that won’t find anything in due time.