In my previous post, I looked at two hormones released during the cephalic phase (gastric secretion that occurs before food is eaten), ghrelin and obestatin, and how they may contribute to runaway eating behavior. Today I’m going to be looking at insulin release during chew and spit (CHSP), a fairly common symptom in eating disorders where the food is tasted, chewed and spit out. Insulin is a small peptide hormone that acts as a key regulator of metabolism; deregulation of insulin signalling plays a role in illnesses such as diabetes and metabolic syndrome. Some people have theorized that CHSP behavior may influence insulin regulation. In fact, there are a number of individuals stating on internet forums that chronic CHSP could lead to insulin resistance, potentially promoting diabetes. As interesting as these theories are, recent data have shown that they are probably not true.
INSULIN RELEASE DURING THE CEPHALIC RESPONSE
The taste of food activates the vagus nerve, a part of the peripheral nervous system that sends signals to the pancreas. In response, the pancreas releases insulin (as well as other hormones) in preparation for food ingestion. This is called the cephalic phase insulin release (CPIR). The magnitude of CPIR has been shown to positively correlate with food palatability, as well as reflect one’s motivation to eat.
The size and duration of CPIR depends on multiple factors. Sweet tasting foods seem to elicit the largest response, on the other hand, multiple studies have shown that sweet tasting solutions or artificial sweeteners fail to stimulate CPIR (although this is controversial). Fatty foods, in combination with sugar, elicits an even larger CPIR. The phase of digestion also matters. In overnight-fasted individuals, CPIR peaks roughly 4min after sensory stimulation and returns to baseline within 10min. After eating (postprandial state), CPIR is much larger and can last up to an hour. Conversely, individual preference towards different food types does not seem to play a role in regulating CPIR magnitude.
Of note is that CPIR is NOT always measured in healthy individuals following presentation and tasting of food. This may be due to the concurrent activation of sympathetic nervous system, which inhibits insulin secretion. Hence individual differences should be taken into consideration when looking at CPIR.
WHAT IS THE PHYSIOLOGICAL ROLE OF CPIR?
Although the magnitude of CPIR is only 1% of total insulin release AFTER a meal, it may play a significant role in metabolism, especially in individuals with metabolic disorders.
After healthy individuals engage in CHSP, CPIR-induced decrease in blood sugar can act as a preemptive measure to prevent a rapid rise in glucose if food is ingested. CPIR also inhibits gluconeogenesis (the generation of glucose from other sources, such as protein), reducing liver glucose production, and inhibits fat-burning in fatty tissue, thereby reducing free fatty acids in the blood.
Remember, CPIR occurs during chewing, but before food is actually swallowed. All of these actions can provide the body with a buffer system for subsequent incoming glucose and fat, and thus help the body maintain metabolic balance (homeostasis). Indeed, CPIR deregulation may be one factor promoting chronic high blood sugar, ultimately leading to Type II diabetes.
CPIR can also indirectly regulate hunger by regulating blood glucose. A decrease in blood sugar in response to CPIR right before food ingestion enhances the state of nutrient deficiency, which promotes greater hunger sensations and increased short-term food intake. (Though, not all studies have found changes in blood glucose levels in response to CPIR.)
CPIR IN PATIENTS WITH ANOREXIA NERVOSA
A study by Teff and Engleman (1996) using the CHSP paradigm demonstrated that in healthy women, attitude towards food influences the magnitude of CPIR, with dietary constraint positively correlating with increased CPIR.
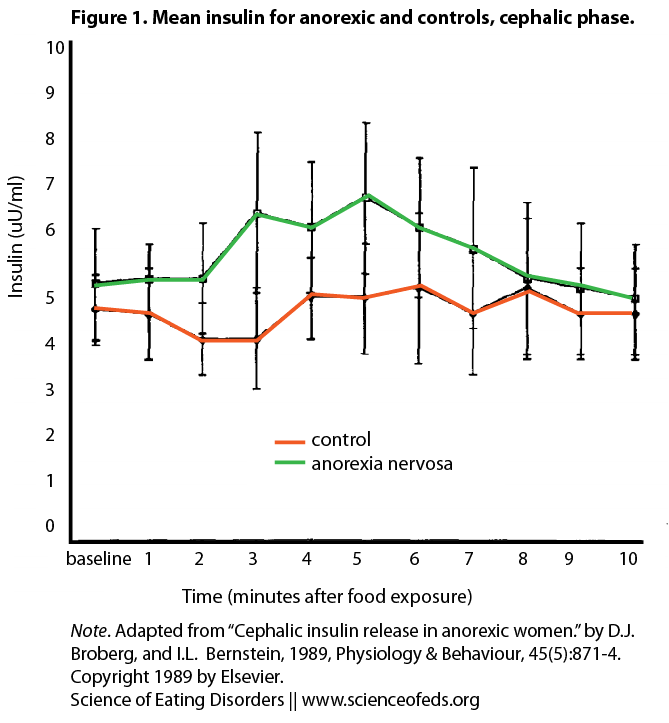
Unfortunately, CPIR is far less studied in patients with anorexia nervosa (AN). In an older study by Brogberg and Bernstein (1989), four AN patients and four age-matched lean controls were presented with an appetizing cinnamon bun (time 0min) after an overnight fast. Blood samples were collected before and after presentation of the food. The subjects were asked to rate the bun’s palatability, and to complete several questionnaires assessing restrained eating behavior. They were then instructed to eat the bun (time 6min).
As you can see, while control subjects did not elicit a significant CPIR, patients with AN showed a 31% increase in insulin levels at 5min compared to 0min, BEFORE food is ingested (in fact one AN subject refused to eat the roll).
Hence, AN patients elicited greater CPIR compared to controls. All subjects rated the bun as equally palatable. Behaviorally, AN subjects scored significantly higher in the item ”cognitive restraint over eating,” and rated lower hunger levels than controls. This psychological self-evaluation directly contradicts the physiological response, which predicts higher levels of hunger. These results support the study by Teff (2001) that attitude towards food and restrictive eating may be the dominating factors towards insulin release during the cephalic phase.
A common notion seen in CHSP forums is that continuous CHSP may lead to diabetes. Based on the above studies this does not seem to be the case. An increased CPIR in AN patients may result in lower blood sugar levels in response to chewing and spitting out food. While this may lead to persistent feelings of hunger, there is no evidence increased CPIR can lead to insulin resistance. However, constant CHSP may cause chronic pulses of insulin release with unknown consequences to the body. Furthermore, the limited number of subjects and studies caution against generalization of the results.
CPIR IN PATIENTS WITH BULIMIA NERVOSA
As CPIR might be related to hunger and overeating, it is conceivable that bulimia nervosa patients, who often show endocrine abnormalities, may have an altered CPIR that, in turn, might be related to the precipitation and maintenance of binges. Moyer et al (1993) conducted one of the few studies examining this hypothesis. They sought to answer 3 questions:
- “Does the profile of CPIR in bulimic women differ from that of non-eating disordered controls?”
- “Does ingestion of food alter subsequent CPIR in bulimics compared with controls?”
- “Are mood state and desire to binge related to CPIR?”
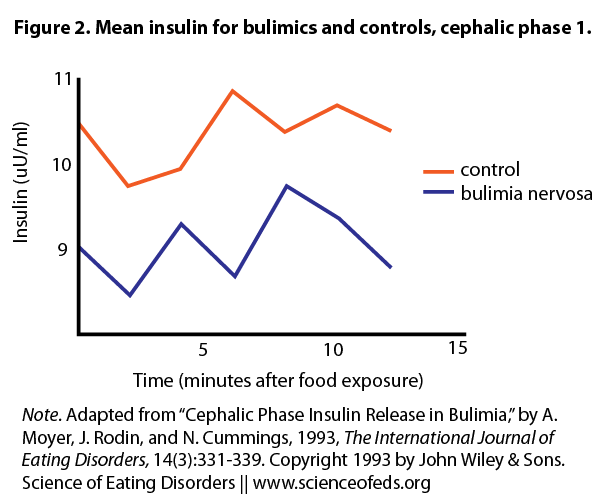
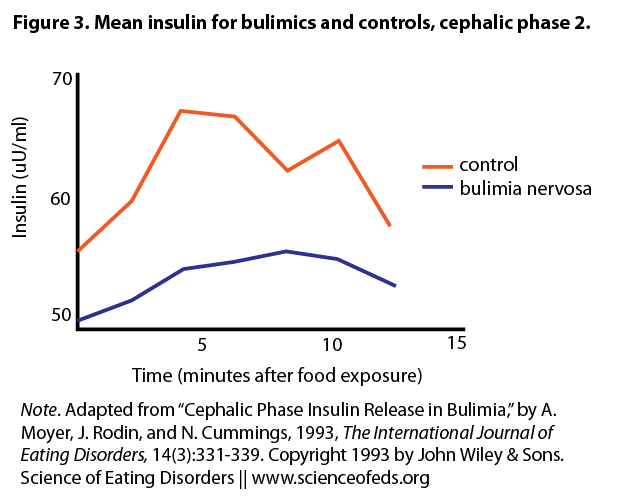
The authors recruited 11 women with BN and 11 age-matched controls. Following an overnight fast, they were instructed to look at, smell, and taste chocolate chip cookies (Trial 1). Although subjects eventually swallowed the food, the authors proposed a 12-minute window before any absorption occurs. Hence insulin changes within 12 minutes can still be viewed as the cephalic “chew” phase. The women were then fed a 350 calorie breakfast, and 40 minutes later (in the postprandial phase) repeated the cookie trial (Trial 2). Recall that in healthy individuals, CPIR is enhanced in magnitude and length of time in the postprandial phase compared to a fasting state.
Under basal conditions, BN subjects had less blood insulin than controls. After the cookie challenge, neither BN nor control group showed an increase in insulin in Trial 1 during the window of the cephalic phase. However, insulin levels for both groups were increased in Trial 2. The authors did not report whether there was a group difference. There was no difference in blood sugar levels in either group in both trials, nor any difference in stress hormone levels. Finally, BN subjects rated higher in the Binge Scale, which correlated with higher scores on depression and anger. However, CPIR was not correlated with mood in either group.
These results strongly suggest that CPIR is elicited in patients with BN, although the magnitude was not compared to healthy controls. Overall, BN and control subjects showed a similar CPIR profiles, both fasting and postprandial. While it’s possible that greater deregulation only occurs during bingeing and purging episodes in BN, there is no clinical data to support this idea.
Hence, although impaired CPIR was hypothesized to contribute to bingeing and purging episodes, these data do not support that notion. In fact, despite reports of neuroendocrine abnormalities, BN patients have normal baseline insulin levels, and show a normal insulin response when ingesting glucose. Hence, it is possible that dysphoric states or stress leads to binge eating, rather than hunger brought on by impaired CPIR.
THE TAKE HOME MESSAGE
Given insulin’s central role in metabolism and its deregulation in metabolic diseases, I had assumed that insulin released during CHSP would have profound effects on the body in both healthy individuals and patients with eating disorders. However, these data do not seem to support this.
In healthy individuals, CHSP results in a rapid rise and fall in insulin secretion, concurrent with a similar change in blood glucose levels. In patients with AN, CPIR is exaggerated, but we don’t know if this corresponds to a drop in blood glucose. One idea is decreased blood sugar may induce physical hunger in patients with eating disorders which eventually overrides any psychological control of eating, leading to more CHSP or food consumption. However there are no studies directly supporting this hypothesis.
In patients with BN, CPIR does not differ significantly from healthy individuals, both under fasting and postprandial conditions. The extent of CPIR seems to correlate with an individual’s attitude towards food, with calorie restriction inducing higher insulin release. While this is proposed to stimulate hunger and feeding, there is no direct evidence to support the hypothesis. Finally, insulin release during CHSP is most likely not directly associated with insulin resistance and diabetes.
However, it’s CRITICAL to keep in mind that studies on this matter are few and far between. The studies I’ve touched on have very few subjects, inadequate controls, and limited measurements of physiological factors. Unfortunately this is an extremely understudied area. A search for “cephalic response” and “anorexia” or “bulimia” on clinicaltrails.gov turned up only 1 result, starting 2007 with no updates since (http://clinicaltrials.gov/show/NCT00493519). Hopefully with increased outreach more health professionals will take interest.
References
Broberg DJ, & Bernstein IL (1989). Cephalic insulin release in anorexic women. Physiology & behavior, 45 (5), 871-4 PMID: 2675143
Moyer A, Rodin J, & Cummings N (1993). Cephalic phase insulin release in bulimia. The International journal of eating disorders, 14 (3), 331-9 PMID: 8275069
Teff KL, & Engelman K (1996). Palatability and dietary restraint: effect on cephalic phase insulin release in women. Physiology & behavior, 60 (2), 567-73 PMID: 8840920
Teff KL (2011). How neural mediation of anticipatory and compensatory insulin release helps us tolerate food. Physiology & behavior, 103 (1), 44-50 PMID: 21256146
Two questions,
You wrote: “CPIR can also indirectly regulate hunger by regulating blood glucose. A decrease in blood sugar in response to CPIR right before food ingestion enhances the state of nutrient deficiency, which promotes greater hunger sensations and increased short-term food intake. (Though, not all studies have found changes in blood glucose levels in response to CPIR.)”
What do you mean not all studies have found this?? I thought CPIR was really well established, no? And if it isn’t, what does that really mean about all the other data (ED data). Or am I misunderstanding something?
Also, why do you think there’s a difference between AN and BN patients? Do you think it is a real difference or just a blip because of the small sample sizes? Any ideas?
Tetyana
CPIR is well established. The thing is, depending on the subject’s state of digestion, CPIR may not lead to an observable decrease in blood sugar levels (decreased blood sugar is generally seen in fasted, not postprandial, subjects). In this case the hypothesis is CPIR acts more to “buffer” the system – to ready it for incoming nutrients that will spike blood sugar so that the body can maintain a relatively stable level of blood sugar.
As for the difference between AN and BN patients, I’m not too sure. Sample size is one thing that needs to be taken into account. However, this study (http://www.ncbi.nlm.nih.gov/pubmed/8032348) also showed relatively normal basal and cephalic phase insulin release in BN patients before and during a meal, so the data seems to be fairly consistent. (They did show persistently high insulin levels after purging though.) It’s possible that disrupted CPIR may only be observed after multiple binge-purge cycles in BN patients.
In the quoted study, the subjects kept a food diary which showed relatively normal macronutrient % before administering the test, and only 6 out of 11 patients engaged in purging the day before the test, which could account for the negative result.
AN patients, on the other hand, consistently show decreased blood glucose, decreased basal and postprandial insulin levels and increased clearance, which is hypothesized as an adaptive mechanism to protect AN patients from hypoglycemia (may account partially for the difference between AN and BN patients, but take it with a grain of salt). So it’s rather surprising in the above study AN patients have INCREASED CPIR.
One factor I’d like to know is the level of insulin RECEPTOR expression in AN vs BN patients. Increased insulin only tells you so much.
Hey Shelly, holdingontoeunoia asked me this question on tumblr and I thought I’d pass it on to you. Thoughts?
“Hey, I’ve been reading your posts about Insulin and we just finished learning about it and its hormonal effects in biology and I have a question. According to the studies you’ve posted, insulin is generally higher in anorectics and lower in bulimics compared to the control, yes? Diabetics also have much lower insulin levels (a graph of insulin levels during the cephalic phase was on our last exam, so this is all fresh in my mind), so would it be unreasonable to hypothesize that the same treatment given to diabetics to regulate insulin levels could be used- or modified- to treat recovering bulimics? And in the case of an anorectic, have medicines with the insulin-specific blockers been developed/ would that also be an option? I’m not completely up to date with ED treatment research or anything, but seeing these posts right after learning about insulin levels has got me thinking about it.”
Heya,
Looks like I didn’t explain the studies too well :P. Insulin release is a tricky issue and I went “full grad student mode”.
So the general conclusion seems to be, compared to controls:
AN patients show LOWER basal insulin levels, increased cephalic phase insulin release, lower postprandial (after eating) insulin levels, and overall low blood sugar.
BN patients, on the other hand, show relatively normal basal, cephalic and postprandial insulin levels if they don’t purge.Blood glucose drops after purging, while insulin levels stays
high for an abnormally long amount of time.
You’re right that Type 2 diabetics have impaired (decreased) CPIR compared to controls (reviewed here: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3056926/. So in some sense AN patients and T2DM patients are “mirror images” of each other. Your idea is really interesting – maybe controlling insulin levels may in turn regulate appetite and eating behaviour. As far as I know there aren’t any trials yet. The major hurdle is PROVING regulating insulin can lead to improvements in eating, or that impaired insulin secretion is a major cause in impaired eating. So far there’s really not enough evidence.
Thanks for the explanation! 🙂 I’m trying to figure out what I want to study and definitely want to head into an eating disorder- related field, but am still trying to decide between becoming a therapist or working in a more medicine-oriented setting. I love this blog and the articles are fascinating, even when they’re a little over my head 😉
It’s a learning process for me too! Not just the science, but also how to communicate it simply and clearly without missing crucial information.
It’s an amazing blog! I’m super honoured to be contributing 🙂
Hi there, this is an interesting study!
I don’t quite understand the figures from the study:
do figures 2 and 3 show that BN subjects generally have lower levels of insulin than controls?
is this somehow related to their purging behaviour? (ie. when their bodies learn over time that some of the food initially ingested is not going to be around for absorption it adjusts insulin release for such?)
Hi there,
Figures 2 and 3 do show that BN subjects have slightly lower insulin levels than controls – however the difference is small and not significant. The authors in this study speculated this is because the BN subjects did not engage in purging behaviour on the day of the test, and because their body weights were within the normal range. This is one of several studies showing that BN subjects may in fact have relatively normal basal insulin levels.
Hope this clears thing up!
Shelly
thanks! yeah it does haha. unfortunately i’ve been doing this quite often although I’m not AN or BN and I was pretty sure there had to be some effect on my body besides the puffy cheeks thing.
but most websites just claim it causes insulin resistance and weight gain, then swing around and claim it causes dangerous weight loss.
nice to see something factual and scientific! (:
🙂
I can’t even say how much I’m glad you have written something like this. I have been diagnosed with AN but have never said about my CHS behavior, in my country there aren’t many people even knowing about such illness as EDNOS…I started food and nutrition studies on University, as I would like to help people like me…
So I was reading only information found on the net and I was terrified by the danger of diabetes, now I’m a bit calmer – I’m impressed by the variety of knowledge you present here! I really admire you!
Hi Alice,
Everything’s been a learning process for me too! Where I come from (originally) EDs in general are not talked about, so it’s great to have this platform to share information and discuss. I do hope more research comes out though – the number of people involved in the studies I talked about is still very few, so it’s hard to draw solid conclusions. Kudos for going into nutrition studies :)!
Hi, I’m really confused does this mean that chewing and spitting doesnt cause insulin resistance , weight gain and diabetes. I’m really worried about the cosequenses of my behaviour. Thank you 🙂
Was wondering if the CPIR levels during the cephalic phase depended on what the subjects injested. Simple or ocmplex carbs, or protein?
Hi Sydnie,
The subjects were given the same meal (chocolate cookie, bun) but CPIR levels is influenced by the macronutrient component. A study in 2007 found that sour, salty, bitter, and umami tasting
treatments did not affect plasma glucose or insulin concentrations (Tonosaki et al. 2007). However, sucrose and saccharin (sweet), did. Finally, starch, a caloric, non-sweet carbohydrate, had no effect on plasma insulin or glucose concentrations. There are also studies that propose CPIR is a learned response to yummy food (Am J Physiol. 1985 Dec;249(6 Pt 1):E639-45.), regardless of macronutrient composition. For example, if you think chocolate is delicious, you might show increased CPIR to coca-containing food, irrespective of its macronutrient composition.
I am really interested in knowing more about the comments you make regarding
‘an individual’s attitude towards food, with calorie restriction inducing higher insulin release.’ Are there any studies that you know of that look at role of restriction in increasing the preferential storage of body fat – I mean as opposed to what we know happens when someone has been restricting and then refeeds, but rather if there is some metabolic signalling that stems from a person with tendencies to AN/ED viewing the food as a threat, and a corresponding shift in body chemistry that drives the body to focus on fat storage. My question is in part in relation to the self-reported calorie intake with women who eat a low level of calories and yet tend to gain body fat over and above what might be their set point. Do we know the mechanism by which attitude affects CPIR? And are there any corresponding treatment protocols in the CBT DBT world that tap into the power of the attitude and work on altering that?
p.s. my interest is in the treatment of AN and R.E.D by changing this ‘attitude’.
I know this is an old post but does this mean I can C/S without weight gain and insulin problems? I understand that it can lead to tooth decay etc but I only C/S savoury food (ham, olives, smoked almonds etc) so the sugar issue isn’t there.
Also, I am an ex smoker so I chew gum all the time, so I don’t quite understand how chewing gum and C/S are that different in terms of activating the stomach to release acid and the mouth to release saliva.
I am currently struggling with an anorexic relapse and do not want to go back to purging on those days where I can’t help but eat “too” much, so I’m trying to figure out what is the lesser of to evils.
Yes, recovery would be the obvious answer but I’m currently watching my kid sister die from a non curative glioma [brain tumour] so we do what we can).
I’m just scared of C/S too much because so many sufferers say they gained weight with C/S, although again, I wonder if it was with a lot more food and food that was sugary rather than 3 mouthfuls of ham.
TIA if you get this and can shed some light on the subject.
lesser of *two evils :p